|
Well, static electricity and our equipment did some things just fine, but our electroscope was recalcitrant, wasn't it! After all was said and done, we were given our assignment for tomorrow:
Assignment: We used ordinary tape to begin to deal with electricity. Electrons were being transferred today!
We celebrated our knowledge of thermodynamics today. Next week, electricity. But for the weekend, R&R!
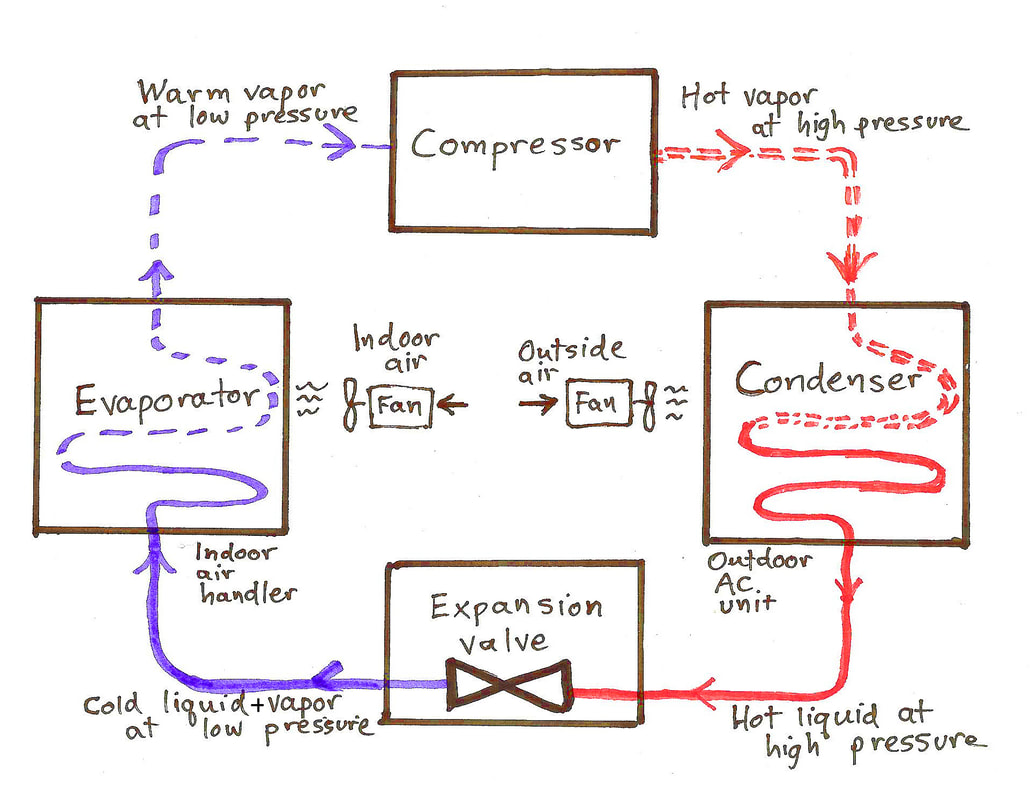
Tomorrow's the Day! Thermodynamics and you! Students asked about the "air conditioner" question that you will deal with. If you go down to Monday's entry, you'll see the diagram that we used in class to describe what happens in a cycle around an AC unit. Click on that diagram or click here to go to a good web page for this purpose. In fact, you'll see the same diagram, because that's where I got it! On that page scroll down to the Question: Q: How does an air conditioner work? In a few paragraphs, the author describes the whole cycle very well. It is a good reference as you practice constructing your own answer to that question. You will have to do that tomorrow. And for those of you who need the solution to Handout Problem 4, here it is:
I'm not saying it's aliens, but...
We are wrapping up our study of heat engines, devices that take in heat and then do some work for us. We'll finish the solutions to our problems tomorrow and deal with questions that students may have. Note that Objectives and Practice Exams are on the Chapter 15 web page. Assignment: Tomorrow we'll turn in our work.
Entropy was our topic today. We noted that it is best thought of as a measure of how widely energy is distributed. Also, entropy has a kind of "half conservation law," namely, entropy can be created, but it can never be destroyed. If you are interested in learning more about this fundamental and essential quantity in science, visit the entropy site hosted by Occidental University. While it's most concerned with a chemistry context, much of what is there is a fine introduction to this unnecessarily mystical quantity. Another rich source of information for high school physics students is the Karlsruhe Physics Course out of the University of Karlsruhe, Germany. Its Thermodynamics unit leads you to a sound understanding of entropy that is much better than the "disorder" idea. It's a novel approach, and a helpful one, if you want to learn more about entropy and its central role in science. Assignment:
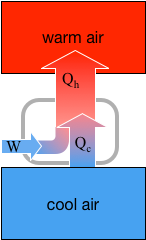
We examined the P-V plot of a diesel engine, just to make some sense about what it tells us. We interpret this P-V plot like any other such plot. We also discussed the efficiency of engines and even mentioned the theoretical maximum efficiency, the "Carnot efficiency." More on that tomorrow. We also dealt with refrigerators. Refrigerators move thermal energy from the cooler interior of the refrigerator to the warmer exterior air around the refrigerator (we call this energy Qc in the diagram below). It takes work to pull off that nifty feat (W), and the energy for that work comes from the electric utility. The total amount of energy that leaves the refrigeration system is equal to that it receives. It is Qh in the diagram. Qh = W + Qc Here's the schematic diagram that we will use to describe how an air conditioning system works: This image is linked to its source, and you will find there a fine description of how air conditioning systems work.
We took a look at some real engines today to get a better idea of how engines work. Here are the links:
We'll take a look at the solutions to our P-V plot problems next week.
|
Physics IIMr. Swackhamer Archives
May 2019
Categories |
||||||||
| Mr Swackhamer's Classes |
|
- Home
- About
- Contact
- Physics I
- Physics II
-
Earth Science
- Earth Science Blog
- Earth Science Chapter 1
- Earth Science Chapter 2
- Earth Science Chapter 3
- Earth Science Chapter 4
- Earth Science Chapter 5
- Earth Science Chapter 6
- Earth Science Chapter 7
- Earth Science Chapter 8
- Earth Science Chapter 9
- Earth Science Chapter 10
- Earth Science Chapter 11
- Earth Science Chapter 12
- Earth Science Chapter 13
- Earth Science Chapter 14
- Earth Science Chapter 15
- Earth Science Chapter 16
- Earth Science Chapter 17
- Earth Science Chapter 18
- Earth Science Chapter 19
- Earth Science Chapter 20
- Earth Science Chapter 21
 RSS Feed
RSS Feed
