|
We crushed a can today, and then we saw a railroad tank car undergo a pressure-related process! It all comes from moving particles. But how can particles condense? One thing we know for sure is that our ideal gas particles cannot stick to each other; they don't attract or repel each other at all!
Assignment:
We finished our discussion of Ideal Gas Law Factors and took a brief look at the Chapter 13 Objectives for the Ideal Gas Law. I have edited them a bit today after school according to our goals this year. The practice test is not long but has many basic ideas in it. Along with our homework and Ideal Gas Model Factors, there is plenty to help you understand the Ideal Gas Model.
Assignment:
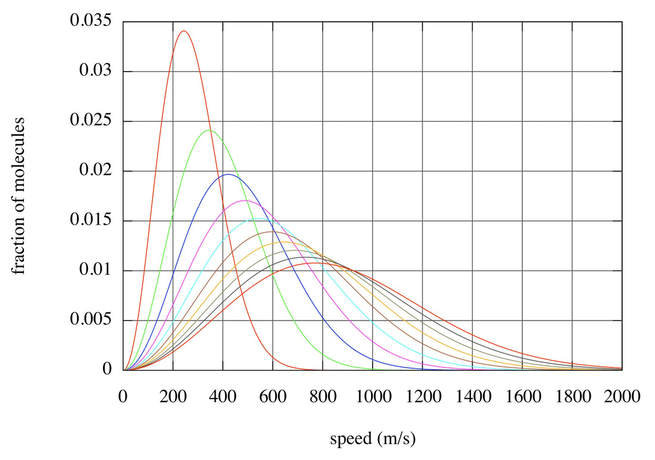
We spend much of our time dealing with the handout from last Friday, Ideal Gas Model Factors. We will finish these problems tomorrow. Also tomorrow, we will have more to say about the Maxwell-Boltzmann speed distribution for the ideal gas model. It has to do with how many particles are moving at a given speed. The distribution shows us what the most common speed is for particles in a gas at a certain temperature. You should also be aware of how the distribution curve changes as the temperature of a sample of gas is raised or lowered. The plot above shows the distributions of the speeds of gas particles for a sample of gas at different temperatures. Which plot is for the sample of gas at its highest temperature? Why do the plots become shorter and wider as the temperature of this gas sample increases?
Assignment:
We used the ideal gas model to deal with some basic questions about gases. In particular, we began class by probing the relationships among the variables that describe ideal gases, namely, pressure, temperature, volume, average kinetic energy, and temperature.
Practice: Ideal Gas Factors After reminding ourselves what STP is and introducing the idea of "root-mean-square" speed, we presented our solution to today's assigned problems: Ch 13 P: 46, 47, 48, 50, 52 One of our preblems today (47) had to do with helium atoms at the 'surface' of the sun. It turns out that today is the first day NASA has received data from the Parker Solar Probe that is going to approach the Sun closer than any other probe. A goal is to find out how the corona gets so hot ( > 1 000 000 K!) even though the 'surface' (aka "photosphere") is just 6000 K. Assignment:
After reviewing some Chemistry and Physics I material, we launched into their implications for the Ideal Gas Model. After a mysterious journey through physical relationships about particles hitting walls, we ended up at a rather startling conclusion: All you need to determine the average kinetic energy of a single ideal gas particle is the temperature of the gas it inhabits.
k is Boltzmann's constant, 1.38 x 10-23 J/K
Assignment:
We considered how to account for pressure, drinking from straws, the effect of temperature on pressure, and other things in terms of a basic particle model of gas. The simulations that we saw today are linked on the Chapter 13 web page. The goal is to help you conceive of particle explanations for phenomena involving matter. Over the next weeks we will use particle models to account for gases, liquids, and solids. Later in the year we will deal with electric conductors and insulators and finally with atomic nuclei. Particle models for matter are at the heart of all these systems, but our models have to have slightly different properties, as we will see, in order to make sense of all that we will observe.
Assignment:
|
Physics IIMr. Swackhamer Archives
May 2019
Categories |
| Mr Swackhamer's Classes |
|
- Home
- About
- Contact
- Physics I
- Physics II
-
Earth Science
- Earth Science Blog
- Earth Science Chapter 1
- Earth Science Chapter 2
- Earth Science Chapter 3
- Earth Science Chapter 4
- Earth Science Chapter 5
- Earth Science Chapter 6
- Earth Science Chapter 7
- Earth Science Chapter 8
- Earth Science Chapter 9
- Earth Science Chapter 10
- Earth Science Chapter 11
- Earth Science Chapter 12
- Earth Science Chapter 13
- Earth Science Chapter 14
- Earth Science Chapter 15
- Earth Science Chapter 16
- Earth Science Chapter 17
- Earth Science Chapter 18
- Earth Science Chapter 19
- Earth Science Chapter 20
- Earth Science Chapter 21


 RSS Feed
RSS Feed
