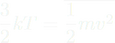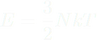Chapter 13: Temperature and Kinetic Theory
Online textbook: Temperature, Kinetic Theory, and the Gas Laws
|
The model: Ideal Gas
PV = NkT Things the Ideal Gas Model can explain:
|
Chapter 13 Objectives
Chapter 13 Ideal Gas Practice Test
Particles, Pressure, Volume, Temperature
Order of treatment of particle models for matter
The rationale for this sequence is that at first gases require only the simplest particle model, the Ideal Gas model. Liquids and solids require what we will call the Many Interacting Particle model, which includes a few more concepts. Therefore, we consider solids and liquids after we deal with how particles account for gases. The flow of ideas is from simpler to more complex. | ||||||||||||||||||||||||||||||
| ch13assign01partmod.pdf | |
| File Size: | 46 kb |
| File Type: | |
- PV = NkT or PV = nRT Understand what each symbol represents and how to do the basic algebra to find one unknown variable.
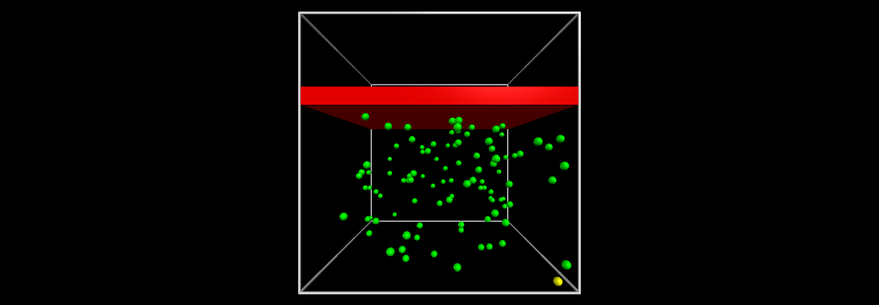
- The four "postulates" of the Ideal Gas model:
- An ideal gas is made of an enormous number of particles with a wide range of speeds.
- These particles are widely spaced (at least on their scale!).
- These particles do not interact except by colliding with each other.
- The collisions of these particles are perfectly elastic.
- You should know the successes and failures of the ideal gas model (i.e. What can it explain, and what can it NOT account for?)
- (3/2)kT = KEavg