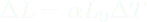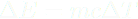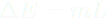Chapter 14: Heat
Online textbook: Heat and Heat Transfer Methonds
Online textbook: Heat and Heat Transfer Methonds
|
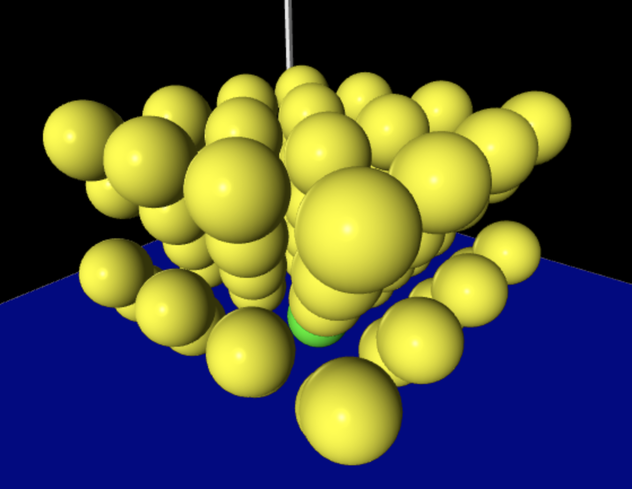
The model: Many Interacting Particles (MIP)
System members:
System properties:
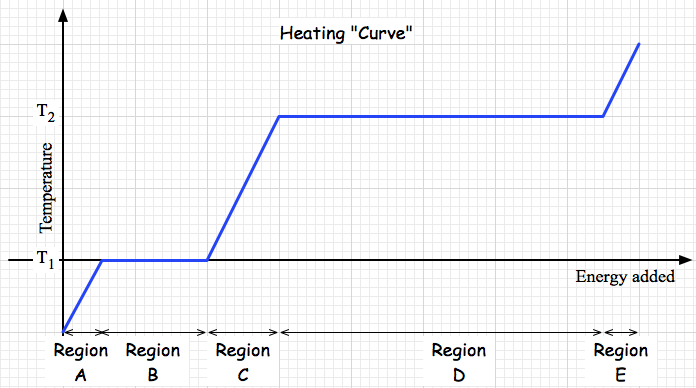
Phase Changes Over what ranges of temperature (on this relative scale) is this system of particles a solid? a liquid? Phases of Water videos Liquid water Ice Steam Ways to represent this system and its interactions
What is going on in each section of this plot?
|
Chapter 14 Objectives
Chapters 13 and 14 Practice Test
WHAT IS A FIELD?
FIELDS AND THE ENERGY THEY STORE It takes energy to build an electric field or a magnetic field, energy that they then possess, and these fields don't disappear without their energy being transferred elsewhere. Energy is conserved, you know! The more these two kinds of fields diminish, the less energy they possess. You can visualize the changing of an electric field and the energy it possesses here. The bars in this simulation represent the energy stored by the red positive and cyan negatively charged particles (kinetic energy) and the electric field (electric potential energy). Just watch this simulation. The bars are color-coded. Click on 'Pause' to see the electric field resulting from the two oppositely charged particles. The field is enhanced when the particles are far apart, and it is diminished when the particles are close together. As the field goes, so goes electric potential energy. Gravitational fields are different; the more energy gravitational fields lose, the stronger they become. The ultimate example of a gravitational field where an enormous amount of gravitational potential energy has been lost is a black hole. Lots of matter has fallen into black holes, resulting in a big loss of GPE and the strongest gravitational fields in the universe. In this chapter we deal with electric fields through which atoms and molecules interact. Latent heats of vaporization and fusion are stored in these electric fields. When oppositely charged particles move closer together, their electric field is diminished, and electric potential energy is lost. Why talk about fields now? The whole point is to help you envision how latent heats of fusion and vaporization are stored. Thermal energy is stored among the motions and oscillations of particles in matter. As you know from Chapter 11, oscillators have both KE and PE. But phase energy (aka "latent heat") is stored in electric fields arising from charge particles separated from each other in that same matter. Phase energy doesn't involve motion or jiggling, and therefore, it doesn't contribute to temperature. Thermal energy involves motion and jiggling and is therefore associated with temperature. Phase energy is not associated with temperature. Temperature arises from motion, including jigging in oscillations. Phase energy is stored in electric fields, not in moving particles. That's why the temperature of boiling water on a stove does not rise above the boiling point when you dial up the burner even higher; all that energy goes into separating the polar water molecules from each other. That energy is stored in the resulting electric fields. Another way to say it is that all that energy goes into building up the electric fields around the water molecules as they separate from each other. So steam at 100˚C has a lot more energy than liquid water at 100˚C, not because it has more thermal energy (it doesn't), but because of all those little electric fields surrounding the separated water molecules. This is the potential energy that humid air has, and it is the source of all the energy that storms exhibit. So humid air at 90˚F has more PE than dry air at 90˚F due to all the separate water molecules and their electric fields, although both have the same thermal energy, which arises from their molecular motions. | ||||||||||||