|
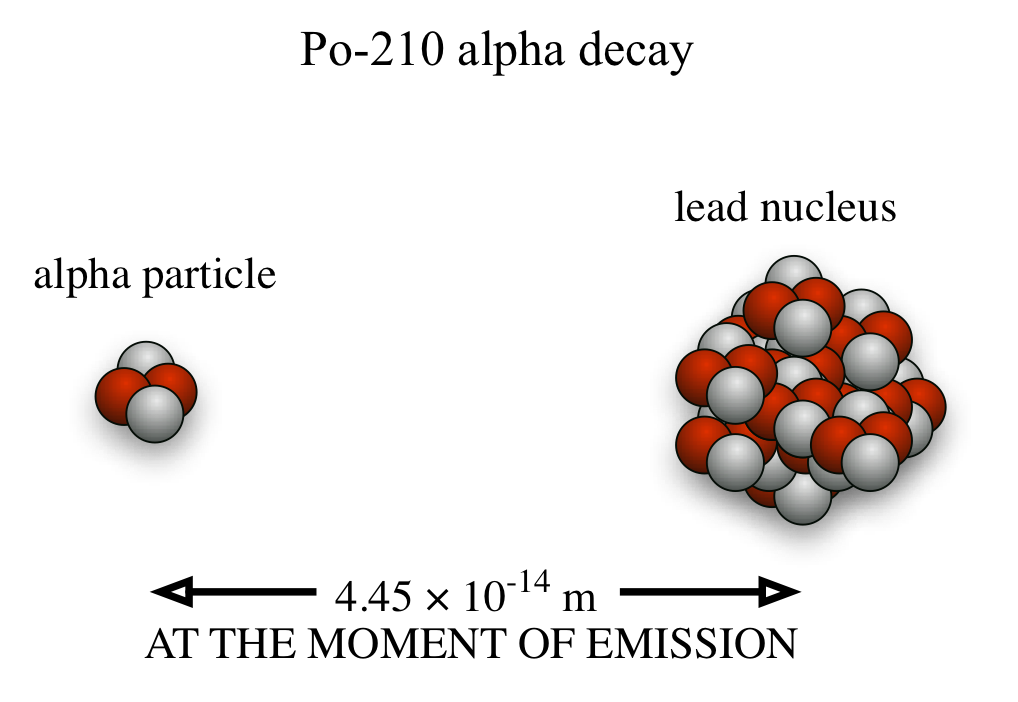
We saw the endpoint of our classical physics today: There is no classical way to explain how an alpha particle exits a nucleus like Polonium-210. While the Po nucleus is only about 8 fm in radius, the alpha particle that leaves it somehow shows up about 45 fm out. How does it just "show up" like that? Classical physics has no answer, but quantum mechanics, which was first worked out by Werner Heisenberg in 1924 or so, describes that this kind of thing can and does happen all the time.
So we have seen two phenomena that cannot be explained by classical physics, the interference of electrons and the "tunneling" of alpha particles out of their parent nuclei. In reality there are many such quantum mechanical phenomena. For example, one can account for the structure of atoms only with quantum mechanics, and that's where all the electron configuration information came from in chemistry. After this interlude we returned to radioactivity by figuring out the isotopes involved in the uranium decay series. Assignment:
Comments are closed.
|
Physics IIMr. Swackhamer Archives
May 2019
Categories |
| Mr Swackhamer's Classes |
|
- Home
- About
- Contact
- Physics I
- Physics II
-
Earth Science
- Earth Science Blog
- Earth Science Chapter 1
- Earth Science Chapter 2
- Earth Science Chapter 3
- Earth Science Chapter 4
- Earth Science Chapter 5
- Earth Science Chapter 6
- Earth Science Chapter 7
- Earth Science Chapter 8
- Earth Science Chapter 9
- Earth Science Chapter 10
- Earth Science Chapter 11
- Earth Science Chapter 12
- Earth Science Chapter 13
- Earth Science Chapter 14
- Earth Science Chapter 15
- Earth Science Chapter 16
- Earth Science Chapter 17
- Earth Science Chapter 18
- Earth Science Chapter 19
- Earth Science Chapter 20
- Earth Science Chapter 21
 RSS Feed
RSS Feed
