|
We are working on understanding the processes of heating up and cooling substances. The goal is to be right at home with phase changes and calorimetry problems.
Assignment:
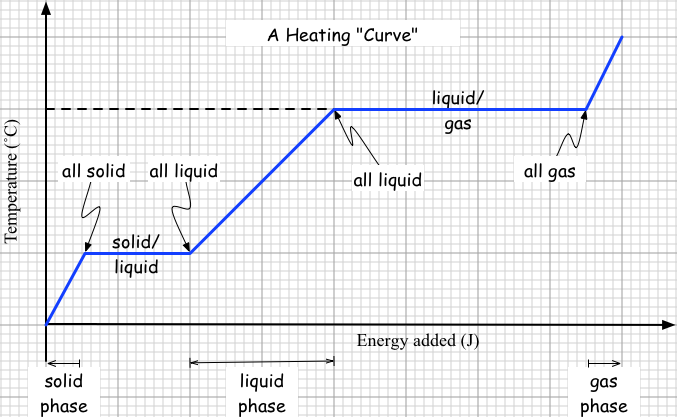
The main thing we dealt with today is how to think about the process of adding energy to some substance to warm it up AND to change its phase. As an example we analyzed warming up 16.5 kg of silver from 20˚C until it is all melted. It turns out there are TWO steps to this process.
2. Melt the silver. This will not change the temperature, because the added energy goes into breaking apart the silver ions from their orderly lattice so tha they can "roll" around, making a liquid. Just to go from a solid at 961˚C to a liquid at 961˚C takes a fair amount of energy: The other example we dealt with is how much energy is needed to take 2 kg of ice from -10˚C to steam at 120˚C. While we didn't do the calculations, we did identify the FIVE steps that have to be accomplished to do this.
Assignment:
The purpose of our experiment today was to determine the heat of sublimation of dry ice. And so we designed our experiments, collected the necessary data, and that just about took the whole period.
Our report will be due on Wednesday, October 17 and should include the usual information:
Our Homework problems, Ch 14 P: 1, 2, 6; 8, 9, 13; and 21, 23, 25 will be due on our SECOND class period after returning from Fall Break. Our dry ice sublimation lab reports will be due on the THIRD class period after Fall Break. That's about it! Enjoy your break! Today we presented our solutions to the assigned problems. While we made good progress, we will not finish our work on them until after Fall Break. We also described an experiment we will do tomorrow. Our goal will be to determine the latent heat of sublimation of dry ice, CO2. .Water was the topic du jour. To get an idea of what is going on with water we observed the three videos linked here: Each phase of water, solid, liquid, and gas, can be described by how particles are bound to their neighbors (or not). There is more potential energy in liquid water than in ice, and there is WAY more potential energy in steam than in liquid water because of the much greater separation of the charged portions of the molecules in steam. (Don't forget, the separation of attracting particles requires energy, and when everything is separated, the expended energy is then stored as potential energy, which we have called "field energy.") The relationship between potential energy and the electric field of attracting particles can be seen in the Bound Particle simulation that we saw. The farther apart the two attracting particles, the greater the potential energy.
Assignments:
The point of today's lesson was to give a reason for the existence of heats of fusion and heats of vaporization. We can think of condensed matter (solids and liquids) as having a thermal energy system and a "phase energy" system. The thermal energy system is made up of all the jiggling particles and contributes to temperature. Through collisions with its particles, the thermal system can gain energy from or lose energy to its environment.
The so-called "phase system" consists of the electric fields enhanced during melting and vaporizong, a process simulated today with our computer simulations. As noted today, these fields take energy to create, and they in fact store this energy. This latent energy is not sensible; it is stored in electric fields, not jiggling particles. When condensation and freezing, particles once again bond, shedding energy to the environment, energy that comes from the now-diminshed elecric fields. The lost latent energy (aka "phase energy" or "field energy") doesn't contribute to temperature, and so there is no change in temperature during a change of phase. The jiggling is just as effective before and after the phase change, which is just another way of saying the temperature doesn't change during the phase change. Assignments:
We considered these questions today:
You should be able to answer these questions in terms of partilcles and the electric field through which they attract each other. Tomorrow we will take a more general look at the fields we encounter in Physics II, namely gravitational fields, electric fields, magnetic fields, and, among nucleons, a "strong" field. Assignment:
|
Physics IIMr. Swackhamer Archives
May 2019
Categories |
| Mr Swackhamer's Classes |
|
- Home
- About
- Contact
- Physics I
- Physics II
-
Earth Science
- Earth Science Blog
- Earth Science Chapter 1
- Earth Science Chapter 2
- Earth Science Chapter 3
- Earth Science Chapter 4
- Earth Science Chapter 5
- Earth Science Chapter 6
- Earth Science Chapter 7
- Earth Science Chapter 8
- Earth Science Chapter 9
- Earth Science Chapter 10
- Earth Science Chapter 11
- Earth Science Chapter 12
- Earth Science Chapter 13
- Earth Science Chapter 14
- Earth Science Chapter 15
- Earth Science Chapter 16
- Earth Science Chapter 17
- Earth Science Chapter 18
- Earth Science Chapter 19
- Earth Science Chapter 20
- Earth Science Chapter 21
 RSS Feed
RSS Feed
