|
Isothermal and adiabatic processes
Assignment:
We turned in our Dry Ice Lab reports.
We discussed problems Ch 13 P: 7, 8, 9, and 10. We also took a first look at the Ch 13 Practice Test. Assignment:
We worked through problem 14 today, and it seems there was good success. Still, I would be surprised if everyone was confident about the way to think about the goings on with energy in this calorimeter system. Be sure to figure out this problem thoroughly.
Assignments:
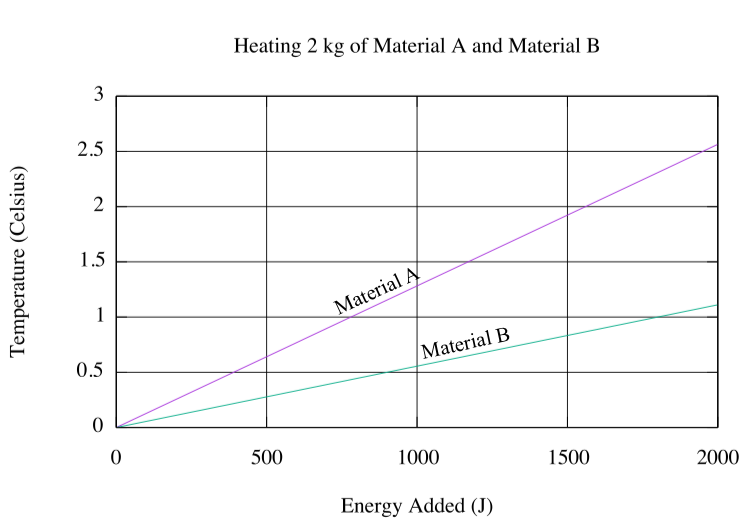
What are the specific heat capacities for Material A and Material B represented in the plot below? Today we dealt with the question of how much energy do we need to heat 2 kg of ethanol from -120˚C to -100˚C. The melting point of ethanol is -114˚C. The specific heat for solid ethanol is 111.5 J/mole K. The standard heat of fusion of ethanol is 4.9 kJ/mole. The specific heat for liquid ethanol is 112.4 J/mole K.
Assignment:
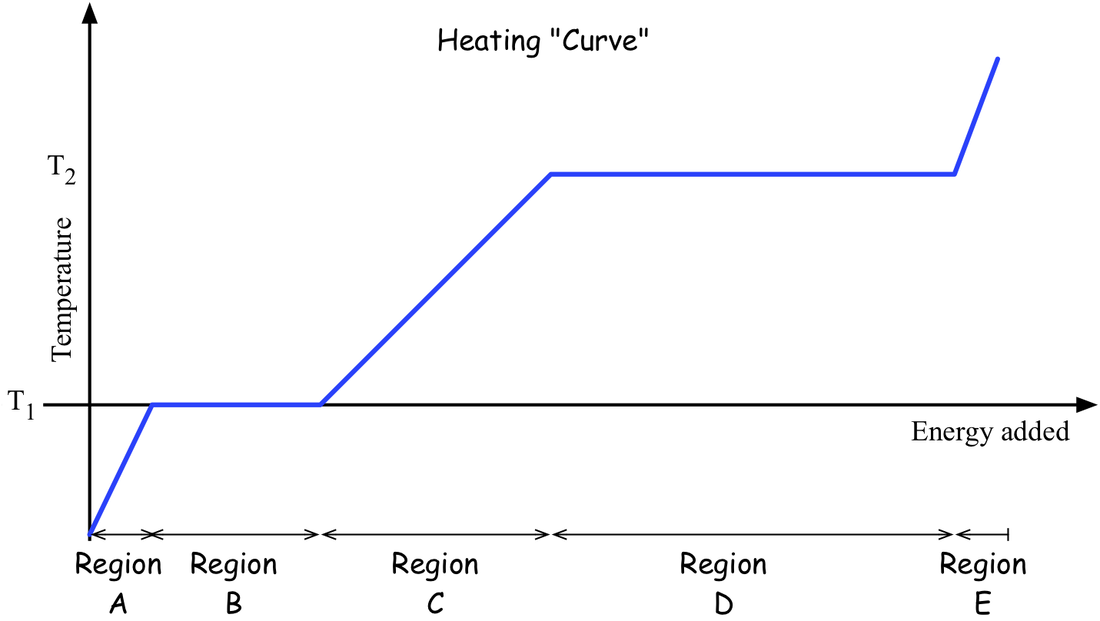
We tackled a so-called 'heating curve' for some generic substance, which in our case consists of straight segments. The question is How do we interpret this? You should be able to describe what is going on with the substance in each of the labeled regions. You should also be able to rank the specific heat capacities for the solid, the liquid, and the gas as represented on this plot.
|
Physics II
Mr. Swackhamer Scottsdale Preparatory Academy Archives
March 2020
Categories |
| Mr Swackhamer's Classes |
|
- Home
- About
- Contact
- Physics I
- Physics II
-
Earth Science
- Earth Science Blog
- Earth Science Chapter 1
- Earth Science Chapter 2
- Earth Science Chapter 3
- Earth Science Chapter 4
- Earth Science Chapter 5
- Earth Science Chapter 6
- Earth Science Chapter 7
- Earth Science Chapter 8
- Earth Science Chapter 9
- Earth Science Chapter 10
- Earth Science Chapter 11
- Earth Science Chapter 12
- Earth Science Chapter 13
- Earth Science Chapter 14
- Earth Science Chapter 15
- Earth Science Chapter 16
- Earth Science Chapter 17
- Earth Science Chapter 18
- Earth Science Chapter 19
- Earth Science Chapter 20
- Earth Science Chapter 21
 RSS Feed
RSS Feed
